Researchers demonstrate non-invasive method for assessing burn injuries
About Optica
30 January 2023
Researchers demonstrate non-invasive method for assessing burn injuries
Neural network based on terahertz spectroscopy data predicts burn healing with 93% accuracy
WASHINGTON — Researchers have developed a neural network model that uses terahertz time-domain spectroscopy (THz-TDS) data for non-invasive burn assessment. They combined the new approach with a handheld imaging device that they developed specifically for fast THz-TDS imaging of burn injuries.
“It is important for healthcare professionals to accurately assess the depth of a burn to provide the most appropriate treatment,” said research team leader M. Hassan Arbab from Stony Brook University. “However, current methods of burn depth evaluation, which rely on visual and tactile examination, have been shown to be unreliable, with accuracy rates hovering around 60-75%. Our new approach could potentially improve the accuracy of burn severity assessments and aid in treatment planning.”
THz-TDS uses short pulses of terahertz radiation to probe a sample. It is being examined for assessing burn injuries because physical changes caused by a burn will produce alterations in the skin’s terahertz reflectivity.
In the Optica Publishing Group journal Biomedical Optics Express, the researchers report the results showing that their artificial neural network classification algorithm can accurately predict the ultimate healing outcome of in vivo burns in an animal study with 93% accuracy. Compared to previous machine learning approaches used by the researchers, the new method reduces the amount of training data necessary by at least two orders of magnitude. This could make it more practical to process big data sets obtained over large clinical trials.
“In 2018, approximately 416,000 patients were treated for burn injuries in emergency departments in the United States alone,” said Arbab. “Our research has the potential to significantly improve burn healing outcomes by guiding surgical treatment plans, which could have a major impact on reducing the length of hospital stays and number of surgical procedures for skin grafting while also improving rehabilitation after injury.”
Better burn assessment
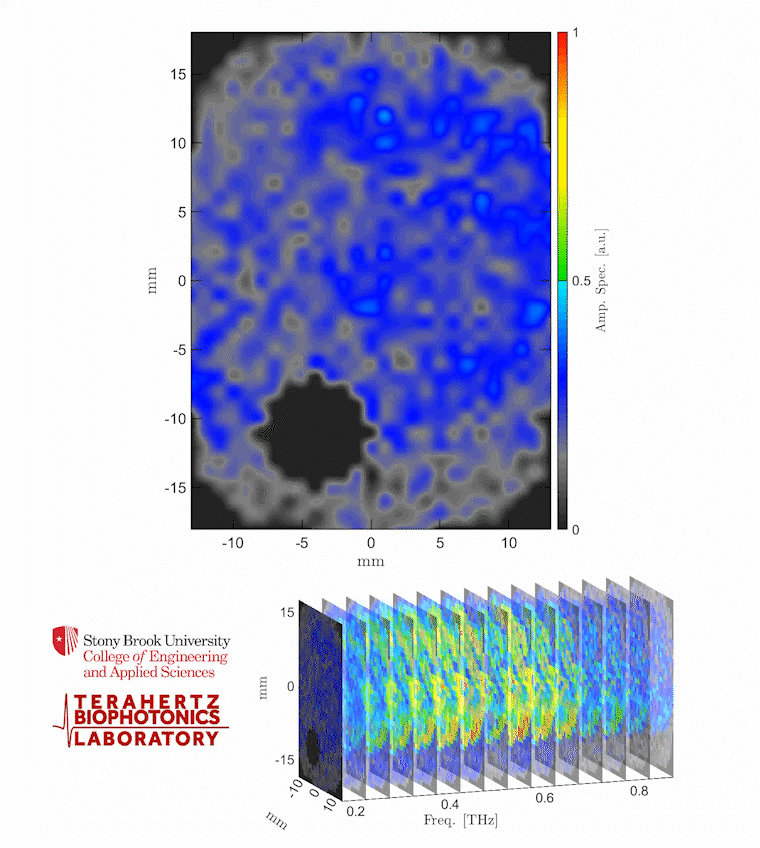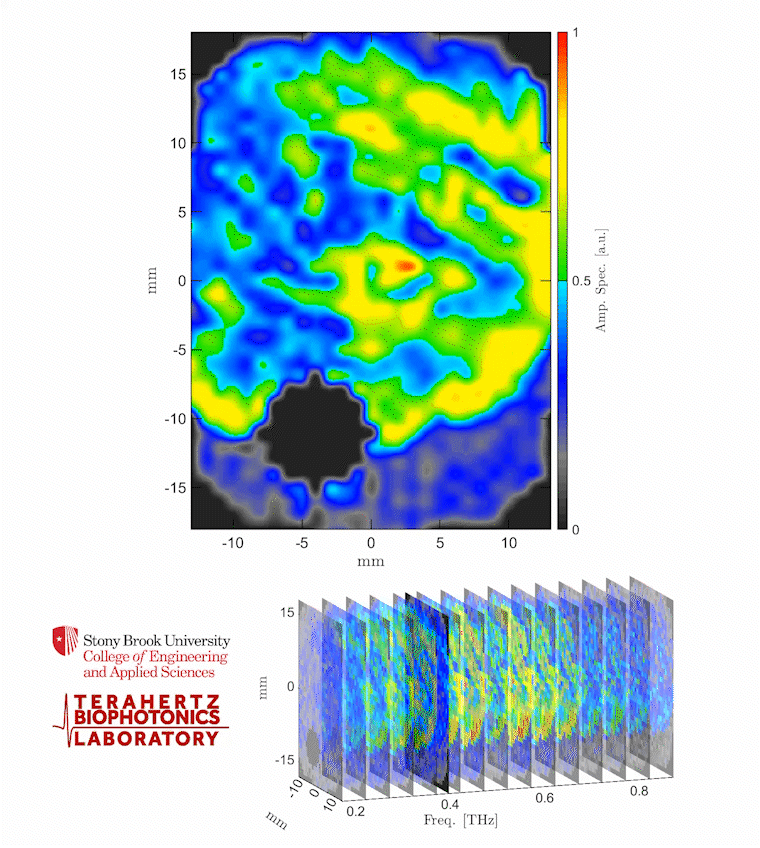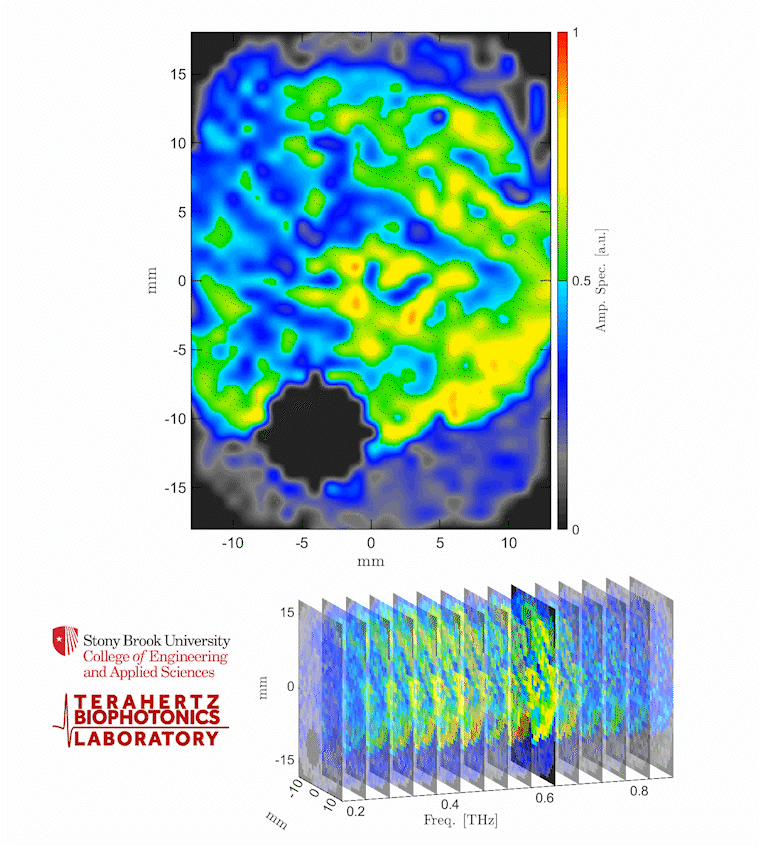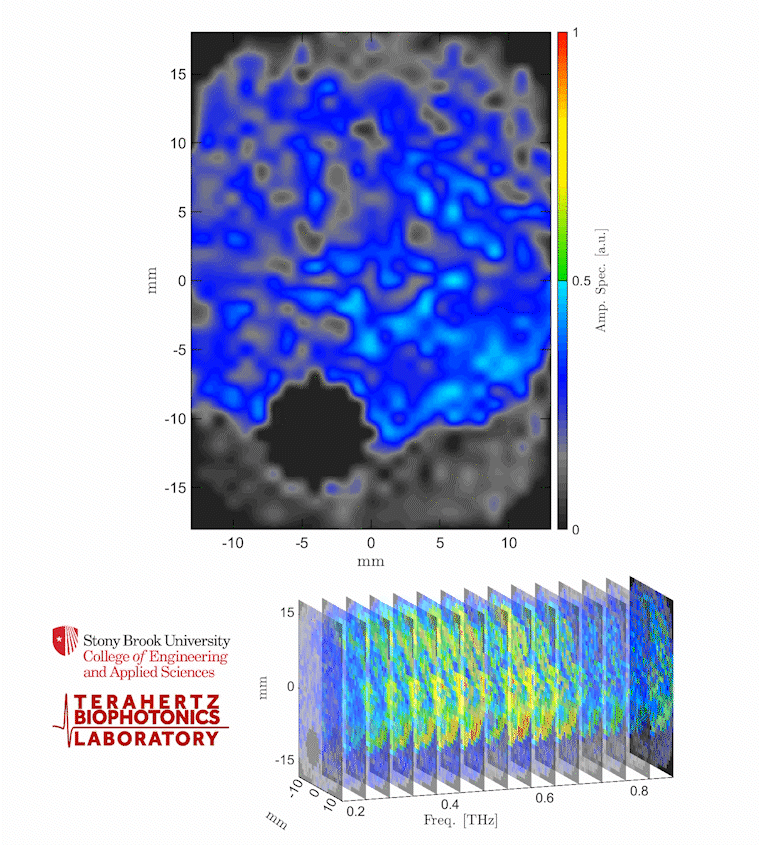
Caption: The video shows a THz spectroscopic image of a burn obtained with the PHASR Scanner by sweeping over a frequency range between 0.2 and 0.9 THz.
Credit: Stony Brook University
Various technologies have been developed to improve burn assessment, but they haven’t been widely adopted in the clinic due to drawbacks such as long acquisition times, high costs and limited penetration depth and field of view. Although THz-TDS looks promising for burn assessment, early demonstrations were limited to point spectroscopy measurements, which don’t account for burn heterogeneity and spatial variations. THz spectroscopy setups also tend to be bulky and expensive and require cumbersome optical alignments, making them impractical for clinical use in real-world settings.
“To address these challenges, we developed the portable handheld spectral reflection (PHASR) scanner, a user-friendly device for fast hyperspectral imaging of in vivo burn injuries using THz-TDS,” said Arbab. “This handheld device uses a dual-fiber-femtosecond laser with a center wavelength of 1560 nm and terahertz photoconductive antennas in a telecentric imaging configuration for the rapid imaging of a 37 x 27 mm2 field of view in just a few seconds.”
Previously, the researchers used numerical methods to extract features from the THz-TDS images and machine learning techniques to estimate the severity grade of in vivo burn injuries using measurements from the PHASR scanner. However, this approach did not consider the physical dynamics and macroscopic changes of the dielectric permittivity of burned skin tissue. Dielectric permittivity describes how a material responds to an electric field.
To investigate the mechanisms that change the complex dielectric function of skin burns in terahertz frequencies, the researchers turned to the double Debye theory, which has been successfully used to explain the interaction of THz radiation with various types of biological tissue.
Predicting severity and healing
“We developed a neural network model utilizing the five parameters obtained from fitting the double Debye model to the dielectric permittivity of burn injuries,” said Arbab. “This physics-based approach allows for the extraction of biomedical diagnostic markers from broadband THz pulses, reducing the dimensionality of THz data for training the artificial intelligence models and improving the efficiency of machine learning algorithms.”
The researchers tested their method by using the PHASR scanner to obtain spectroscopic images of skin burns and measure the permittivity of the burns. After determining the Debye parameters, the researchers used this data to create a neural network model based on labeled biopsies. The model estimated the severity of the burns with an average accuracy rate of 84.5% and predicted the outcome of the wound healing process with an accuracy rate of 93%.
The researchers note that clinical testing of both the technique and the handheld imaging device are needed before this technique could be integrated into the existing workflow of clinical burn assessment.
The research reported in this study was supported by the National Institute of General Medical Sciences of the National Institutes of Health under award number R01GM112693.
Paper: M. E. Khani, Z. B. Harris, O. B. Osman, A. J. Singer, M. H. Arbab, “Triage of in vivo burn injuries and prediction of wound healing outcome using neural networks and modeling of the terahertz permittivity based on the double Debye dielectric parameters,” Biomed. Opt. Express, volume 14, issue 2, pp. 918-931 (2023).
DOI: doi.org/10.1364/BOE.479567
About Optica Publishing Group
Optica Publishing Group is a division of the society, Optica, Advancing Optics and Photonics Worldwide. It publishes the largest collection of peer-reviewed and most-cited content in optics and photonics, including 19 prestigious journals, the society’s flagship member magazine, and papers and videos from over 1200 conferences. With over 520,000 journal articles, conference papers and videos to search, discover and access, its publications portfolio represents the full range of research in the field from around the globe.
About Biomedical Optics Express
Biomedical Optics Express serves the biomedical optics community with rapid, open-access, peer-reviewed papers related to optics, photonics and imaging in biomedicine. The journal scope encompasses fundamental research, technology development, biomedical studies and clinical applications. It is published monthly by Optica Publishing Group and edited by Ruikang (Ricky) Wang, University of Washington, USA. For more information, visit Biomedical Optics Express.
Media Contact
